
Not deflected in the way that would be expected of electrically charged When the German physicist Heinrich Hertz passed the rays through an electricįield created by metal plates inside a cathode ray tube, the rays were When physicists movedĪ magnet near the glass, they found they could push the rays about. Were needed to resolve the uncertainties. It ought not to be difficult to discriminate between views so different, yet experienceĬase." - J.J. Thomson, thought that all material particles themselves might be some kind of structures built out of ether, so these views were not so far apart.Īre held as to these rays. Maybe cathode rays were similar to light waves? Another possibility was that cathode rays were some kind of material particle. 
Hat could these rays be? One possibility was that they were waves traveling in a hypothetical invisible fluid called the "ether." At that time, many physicists thought that this ether was needed to carry light waves through apparently empty space. Evidently some kind of ray was emitted by the cathode and lighting up the glass. In 1859 a German physicist sucked out still more air with an improved pump and saw that where this light from the cathode reached the glass it produced a fluorescent glow. and the interior of the tube would glow in lovely patterns. They took a glass tube with wires embedded in opposite ends. Thomson,Ĭience lecturers who traveled from town to town in the mid nineteenth century delighted audiences by showing them the ancestor of the neon sign. Us so promising an opportunity of penetrating the secret of electricity." - J.J. By incorporating engaging teaching techniques like experiments, visual aids, and real-life applications into your lessons, you can inspire students’ interest in atomic theory and pave the way for future discoveries."There is no other branch of physics which affords Teaching students about Rutherford’s planetary model is a valuable lesson in scientific curiosity and perseverance. Relate to Real-Life Applications: Explain how advancements in understanding atomic structure have contributed to innovations in numerous fields such as medicine, technology, and energy production. Visual Aids: Employ various visual aids, such as diagrams, animations, and interactive software programs, so students can better visualize atomic structure. Promote critical thinking by letting them debate on potential shortcomings of the model, which will later pave the way for understanding Niels Bohr’s improvements to atomic theory. Group Work and Discussion : Encourage students to work in groups to explore different aspects of the atomic model. Conduct a scaled-down version of the experiment in the classroom or utilize a virtual simulation to help students understand how Rutherford’s observations helped him form his hypothesis. Hands-On Experiments : Discuss the Gold Foil Experiment that led Rutherford to propose his model. 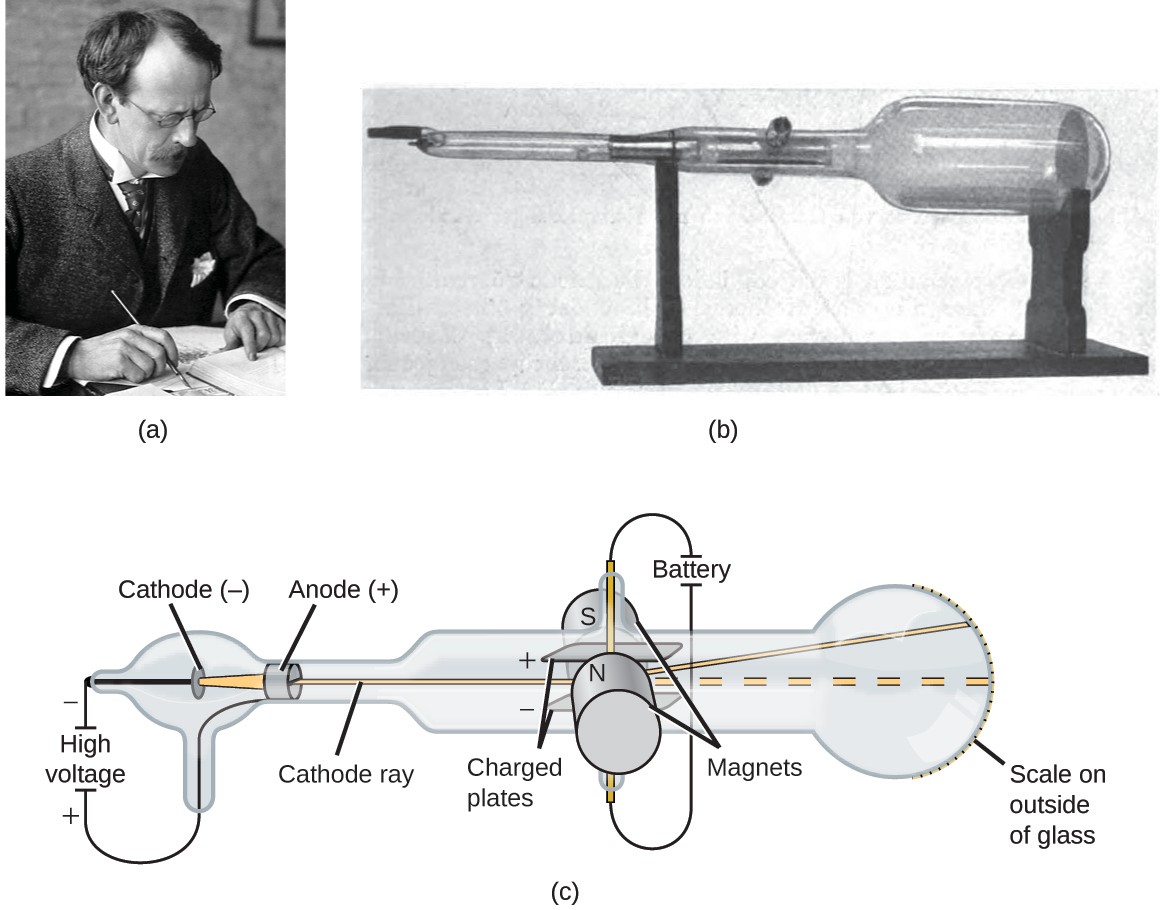 
For example, compare Rutherford’s planetary model to our solar system, with the nucleus representing the sun, and electrons orbiting as planets. Use Analogies: Use relatable analogies to help your students grasp complex concepts easily. Thomson’s “plum pudding” model and explain how Rutherford’s findings fundamentally changed our perspective on atoms. Highlight previous theories on atomic structure and use visuals or videos to showcase J.J. To make learning about the Rutherford planetary model engaging and enjoyable for students, consider adopting these innovative teaching techniques:Ĭontextualize : Begin by providing historical context to help students appreciate the significance of Rutherford’s contributions. The planetary model shaped modern chemistry, allowing scientists to delve further into quantum mechanics and lay the foundations for today’s understanding of atoms. After conducting his experiments, however, Rutherford disproved this model and provided valuable insights into atomic structure. Thomson’s “plum pudding” model was widely accepted. This model revolutionized our understanding of atomic structure by proposing that atoms consist of a dense, positively charged nucleus surrounded by negatively charged electrons orbiting in nearly empty space.īefore Rutherford’s discovery, J.J. Understanding the Rutherford Planetary ModelĮrnest Rutherford, a New Zealand physicist, presented his popular atomic model known as the “Rutherford Planetary Model” after conducting the famous Gold Foil Experiment. In this article, we will explore effective methods for teaching students about the Rutherford planetary model and how to inspire their curiosity for atomic theory. One of the most important scientific discoveries was made by Ernest Rutherford in 1911 with the development of his planetary model of the atom. Over centuries, humankind has strived to better understand the universe, constantly questioning and refining our understanding of the world around us. The beauty of science lies in its never-ending quest for knowledge.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
